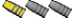After rendering 360 lbs of lead as discussed and pictured in a very recent thread, the cast iron pot was left with a thin layer of lead at the bottom that could not be removed with a sharp scraper or steel wool. Over the years I had run across many threads suggesting that a 50/50 solution of hydrogen peroxide and vinegar, common items in most households, would remove lead from firearm bores. I had researched the subject to some extent and found that the solution results in paracetic acid, a corrosive agent known to etch iron and steel in strong concentrations.
But now the leaded pot was the ideal test sample for a lead cleaning test with the solution. I took it a step further as detailed below. So, after scraping the heavier layers of lead and crud out of the bottom of the pot, I scrubbed it with steel wool. A splotchy thin layer of lead remained, filling the pores and covering most of the bottom. Next I poured in a pint of 3% hydrogen peroxide and a pint of 5% acidic vinegar. Within a few minutes the solution started turning rust red. In about an hour approximately 75% of the lead was gone and the solution seemed to have stopped working. So I dumped out the old solution, wiped the residue out of the bottom of the pot and added a fresh solution of equal parts hydrogen peroxide and vinegar. As before, the solution started to turn rust red but within 15 minutes all the remaining lead was gone.
Based on the results it was clear to me the paracetic acid was not only dissolving the lead but consuming the surface layer of the iron by turning it into iron oxide (rust), hence the deep rust red coloration of the solution. It was also clear that, based on the strength of the ingredients the acid only lasted so long as it was used up in the process of dissolving the lead and creating iron oxide. Since the solution was clearly etching the iron my next step was to figure out some way to quantify the rate of etching.
So I cut off a hunk of clean #0 steel wool, put it in a glass jar and covered it with the solution. The wool was pressed down to remove most of the air and it settled to the bottom. The steel wool + solution immediately began to turn red and create bubbles/gas. After an hr the solution seemed to have stopped working. The steel wool was removed, cleaned under hot water and inspected. The strands were now much finer, closer to #0000 steel wool.
At the same time the steel wool was immersed I grabbed a short piece of iron flat stock, buffed the surface with a wire wheel and stuck it in a glass jar, partially filling the jar with the solution. The solution slowly started to turn red. The photos below are the before and after results of the solution on the iron flat stock after 1 hr. Although the lighting is not the same for both photos and I could not feel the difference in the two surfaces by rubbing a finger over them, it�s clear the iron surface was etched.
The bottom line is a solution of hydrogen peroxide and vinegar will never be used in my firearms to remove lead. Some contributors to the forum threads on the subject suggest is OK to use the solution for a few minutes, and other report allowing it to soak in the bore overnight. Based on my findings even short term repeated use will most definitely etch the bore to some degree.
Wayne
[URL=http://s47.photobucket.com/user/RealTexasMac/media/Hydrogen%20Peroxide%20amp%20Vinegar_zps0vkbe6zc.jp g.html][IMG]http://i47.photobucket.com/albums/f165/RealTexasMac/Hydrogen%20Peroxide%20amp%20Vinegar_zps0vkbe6zc.jp g[/IMG][/URL]

|
   
   
|
|



 Reply With Quote
Reply With Quote DougGuy
DougGuy