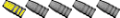Two subjects on oxidation.
How to people prevent plain lead ingots from oxidizing in storage? I have some pucks that have been in a crate in the garage for several years and they have either a thicker black oxide coating or white oxide dust coating. Planning to cast this stuff into larger 2.5 lb ingots using the ingot molds from the group buy / vendor Lakehouse.
I does seem to be an issue for plain lead and not much of an issue for lead with tin alloy.
I was smelting some old lead sheathed underground telephone cable. Full of paper wrapped thin wires. I melted it over a fire in the back yard since I was burning some scrap wood. I also figured the wire covering would smoke and might have been plastic so outside activity.
The cable was somewhat heavily tarnished and it seems to me it was really hard to get the lead clean of oxides and flecks of "stuff" Flux with sawdust, stir, skim. Two ingots ladled out and it had to be skimmed again. Stirring brought more stuff up. It was only 20# of lead but I don't recall having this much trouble getting and keeping lead clean. Eventually I had the stuff in nice ingots but wondered if maybe I could have used some other approach that would have worked better.
I did wonder if the burned paper wire wrapping ash got infused in the melt. It seemed to turn to a black ash.
The cable only yielded 22# of lead but it also had 7.5 lbs. of copper wire that should yield about $13 so almost paid for the purchase.

|
   
   
|


|





 Reply With Quote
Reply With Quote